
EAU 2021: DANUBE Post-Hoc Analysis: Outcomes for Durvalumab with or Without Tremelimumab by Cisplatin Eligibility and PD-L1 Biomarker Status in Metastatic Urothelial Carcinoma

EAU 2021: DANUBE Post-Hoc Analysis: Outcomes for Durvalumab with or Without Tremelimumab by Cisplatin Eligibility and PD-L1 Biomarker Status in Metastatic Urothelial Carcinoma
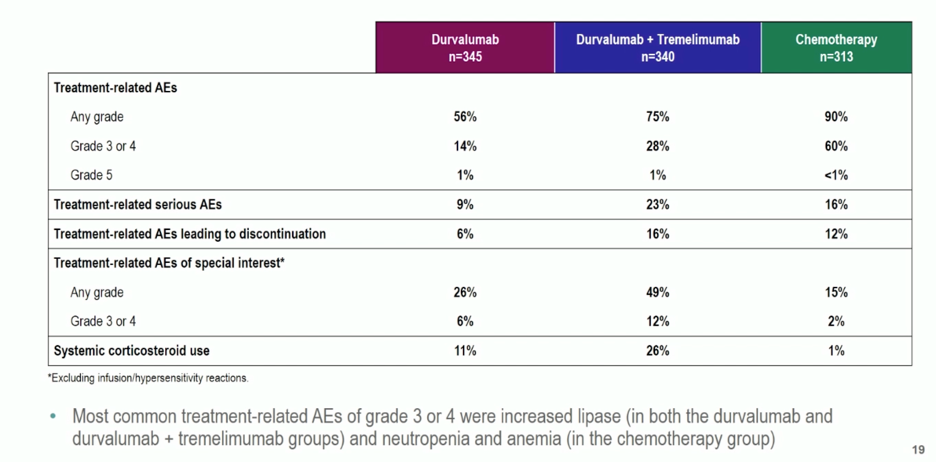
ESMO Virtual Congress 2020: A Phase 3, Randomized, Open-Label Study (DANUBE) First Line Durvalumab with or without Tremelimumab vs Standard of Care Chemotherapy in Patients with Unresectable, Locally Advanced or Metastatic Urothelial

AstraZeneca Reports Results of Imfinzi and Tremelimumab in P-III DANUBE Study for Unresectable Stage IV

EAU 2021: DANUBE Post-Hoc Analysis: Outcomes for Durvalumab with or Without Tremelimumab by Cisplatin Eligibility and PD-L1 Biomarker Status in Metastatic Urothelial Carcinoma

Cabozantinib Plus Durvalumab in Patients With Advanced Urothelial Carcinoma After Platinum Chemotherapy: Safety and Preliminary Activity of the Open-Label, Single-Arm, Phase 2 ARCADIA Trial - Clinical Genitourinary Cancer

EAU 2021: DANUBE Post-Hoc Analysis: Outcomes for Durvalumab with or Without Tremelimumab by Cisplatin Eligibility and PD-L1 Biomarker Status in Metastatic Urothelial Carcinoma

Continued) Key clinical trials' efficacy data of ICIs in patients with... | Download Scientific Diagram

Andrea Apolo, M.D. on Twitter: "Nice summary of ongoing phase 3 randomized #immunotherapy clinical trials in 1st line #bladdercancer #GU18 https://t.co/roifTmpolW" / Twitter

Is it Time to Consider Eliminating Surgery from the Treatment of Locally Advanced Bladder Cancer? - European Urology

Assessing the magnitude of reporting bias in trials of homeopathy: a cross-sectional study and meta-analysis | BMJ Evidence-Based Medicine

Clinical evidence for the first-line treatment of advanced urothelial carcinoma: Current paradigms and emerging treatment options - Cancer Treatment Reviews

ESMO Virtual Congress 2020: A Phase 3, Randomized, Open-Label Study (DANUBE) First Line Durvalumab with or without Tremelimumab vs Standard of Care Chemotherapy in Patients with Unresectable, Locally Advanced or Metastatic Urothelial

Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open-label, multicentre, phase 3 trial - The Lancet ...

ESMO Virtual Congress 2020: A Phase 3, Randomized, Open-Label Study (DANUBE) First Line Durvalumab with or without Tremelimumab vs Standard of Care Chemotherapy in Patients with Unresectable, Locally Advanced or Metastatic Urothelial

ESMO Virtual Congress 2020: A Phase 3, Randomized, Open-Label Study (DANUBE) First Line Durvalumab with or without Tremelimumab vs Standard of Care Chemotherapy in Patients with Unresectable, Locally Advanced or Metastatic Urothelial

EAU 2021: DANUBE Post-Hoc Analysis: Outcomes for Durvalumab with or Without Tremelimumab by Cisplatin Eligibility and PD-L1 Biomarker Status in Metastatic Urothelial Carcinoma

Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open-label, multicentre, phase 3 trial - The Lancet ...
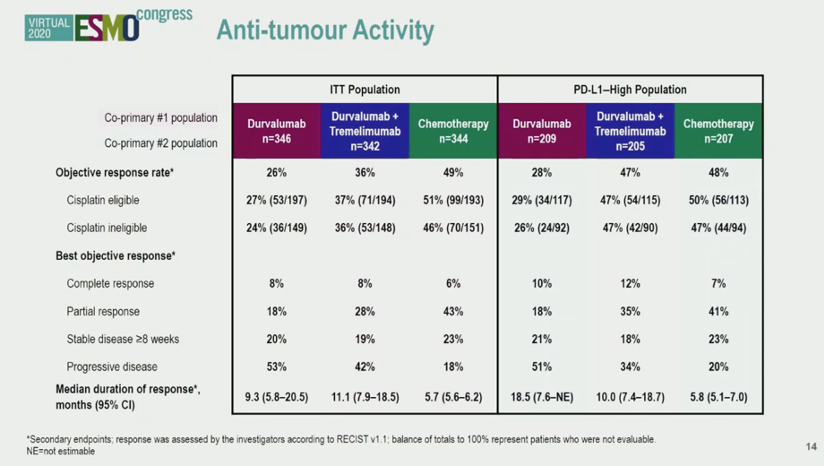
ESMO Virtual Congress 2020: A Phase 3, Randomized, Open-Label Study (DANUBE) First Line Durvalumab with or without Tremelimumab vs Standard of Care Chemotherapy in Patients with Unresectable, Locally Advanced or Metastatic Urothelial

Imfinzi (Durvalumab) a New PD-L1 Inhibitor Approved for the Treatment of Advanced or Metastatic Urothelial Cancer

Primary results of STRONG: An open-label, multicenter, phase 3b study of fixed-dose durvalumab monotherapy in previously treated patients with urinary tract carcinoma - ScienceDirect





